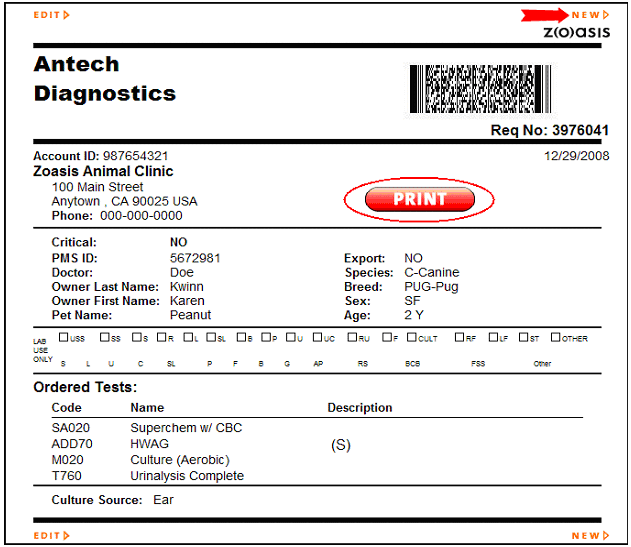
40 lab requisition form with labels
HCG - Serum, Quantitative - UI Health Care Label Mnemonic: HCG : Epic code: LAB142: Order form: Laboratory Requisition: Specimen(s): Plasma. Collection Medium: Plasma Separator Tube 4.5 mL: Alternate Collection Media: Call laboratory for additional acceptable specimen collection containers. ... This assay recognizes the intact HCG "holo-hormone" produced in pregnancy as well as other ... Clarity Labs - Clarity Labs If you are a patient who had COVID-19 testing performed by Clarity Labs, please enter the requisition number, last name, first initial and the date of birth below to view a copy of your result report. View COVID-19 Results NEWSROOM Visit the Center for Disease Control to get the latest news updates about COVID-19. Visit Site About Clarity Labs
Label Submission and Approval System (LSAS) | Food Safety and ... Who do I contact if I need assistance or have a technical issue concerning LSAS? The LSAS administrator will be your first contact. Email the administrator at LSAS@usda.gov or call 301-504-0837. We ask that you use these resources first rather than calling the main Labeling and Program Delivery Division (LPDD) lines so that we can track all issues and provide appropriate resolutions.

Lab requisition form with labels
Procedures for Requisition Entry and Purchase Orders Purchase Orders. Procurement receives an approved requisition and creates a purchase order based on the information entered on the requisition. The requester receives an email with a copy of the purchase order. Procurement issues the purchase order to the vendor to place the order. Syphilis Treatment Follow-Up (RPR with Titer) Diagnostic Laboratories (UIDL) Test Directory 319-384-7212 (local) 1-866-844-2522 (toll free) ... Epic code: LAB8648: Order form: Laboratory Requisition: Specimen(s): Serum. Collection Medium: BD Gold SST 5 mL Vacutainer: Minimum: 5 mL serum from a BD Gold SST tube. Rejection Criteria: ... Place completed requisition into outside pocket of bag ... N.C. DPH: State Laboratory of Public Health Interim Guidance Advisory: Pursuant to Session Law 2020-4 Section 4.10.(a), the Department of Health and Human Services will be requiring each person in charge of a laboratory providing diagnostic service in this State and any other health care provider licensed in this State that provides diagnostic service to report the results of all COVID-19 testing to the Department of Health and Human ...
Lab requisition form with labels. At-Home STD Tests: Everything You Need to Know - Healthline At the testing center, present your lab requisition form. You won't be required to provide identification. A healthcare professional, such as a nurse, will take the required sample. Forms and Guidelines - School of Veterinary Medicine Forms and Guidelines. A Clinical Laboratory Sample Submission form (fillable pdf) must be included with each laboratory submission. Clearly indicate which test(s) are requested, as listed in the test menu. Please note: Certain tests require alternate submission forms, listed below by laboratory. Forms & Submission Requirements | FDA ANDA Forms and Submission Requirements. Drug Master Files (DMFs) DMF Submission Resources. Content current as of: 02/15/2022. Regulated Product(s) Drugs; Forms & Submission Requirements. When do lab requisitions expire? Explained by FAQ Blog The test requisition combines patient registration information, billing information, specimen information, barcoded specimen labels and a provider order for confirmation of testing. The patient bill requisition should be completed by the provider or authorized staff and signed by the ordering provider. What is blood test EUC?
List of California Proposition 65 Product Testing Companies After you submit a requisition form for your products' CA prop 65 testings, they will perform the test and, as long as your product is compliant, they will draft a test report for your products. Also, they offer an online platform that allows clients to track the test status anytime. Services Product testing Product certification Factory inspection Guidance for acceptance of cervical screening samples in laboratories ... All required fields of the HMR101 form must be completed correctly. 6.1 Essential specimen data requirements Required data are: the patient's full name (at least first name and surname) 2 other... Laboratory Safety: Free Checklists & PDF Reports | SafetyCulture Proper labels Labels are important elements of laboratory safety. They help in identifying chemicals, its substance, and the hazards associated with it. A proper label should have the name of the substance, its concentration, the date it was received, and the name of the person responsible for it. Record of an incident Calibration Forms: Top 3 [Free Download] - SafetyCulture What is a Calibration Form? A calibration form is used by technical services teams in the maintenance of functionality, quality, and accuracy of measuring equipment. It helps ensure that workers adhere to calibration standards set by the organization to meet ISO 9001:2015 quality audit requirements. Calibration Form
N.C. DPH: State Lab > Forms Patient Request Form to Obtain Completed Laboratory Results Two forms are required for the NCSLPH to be permitted to release laboratory results to patients. The NCSLPH is not responsible for interpreting laboratory test results. The HIPAA Authorization Form (PDF, 69 KB) grants the NCSLPH consent to distribute patient results. Bronson Animal Disease Diagnostic Laboratory (BADDL) Contact the Lab Reddy Bommineni, DVM, PhD, DACVM, DACPV Chief, Bureau of Diagnostic Laboratory Email: DiagLab@FDACS.gov (321) 697-1400 (321) 697-1467 Fax Hours and Location Hours of Operation Monday-Friday 8 a.m.-5 p.m. Closed on state holidays. Delivery and Service Address 2700 N. John Young Pkwy. Kissimmee, FL 34741-1266 Fill - Free fillable Laboratory Request Form PDF form Use Fill to complete blank online OTHERS pdf forms for free. Once completed you can sign your fillable form or send for signing. All forms are printable and downloadable. The Laboratory Request Form form is 1 page long and contains: Fill has a huge library of thousands of forms all set up to be filled in easily and signed. COVID-19 | Wisconsin State Laboratory of Hygiene Packaging and Shipping: If you have questions about how to submit specimens for testing, contact WSLH Customer Service at 800-862-1013. Submit specimens with a completed WSLH COVID-19 Requisition Form.If your laboratory doesn't already have this form, please call WSLH Clinical Orders at 800-862-1088 during normal business hours (Monday - Friday 7:45 AM - 2:45 PM).
FSIS Compliance Guideline for Label Approval | Food Safety and ... FSIS Compliance Guidance for Label Approval. Replaces: August 2017 version. This guidance document provides official establishments information about the types of labels that must be submitted to LPDS for approval. Included are specific examples of special statements and claims that must be submitted to LPDS for approval. This guideline relates ...
DD Forms 1000-1499 - Washington Headquarters Services DD1434. United Kingdom (UK) Customs Declaration for the Importation of Personal Effects of U.S. Forces/Civilian Personnel on Duty in the UK. 5/1/2015. No. A&S. DD1435. COMSEC Maintenance Training and Experience Record. 5/1/2009. No.
Midwest Laboratories The mission of Midwest Laboratories is to drive a positive impact on the lives of our employees, clients, and communities. Midwest Laboratories is an ISO 17025 and NELAP accredited lab. Operating a 14-building campus headquartered in Omaha, NE. Choose from supported industries below to view related documents and available test packages, or view ...
University of Washington Laboratory Medicine - Patient Care A requisition form must accompany specimen(s) sent to the laboratory. The patient's name and hospital number on the requisition form must match name and number on specimen container. When a discrepancy exists, specimens are unacceptable and the policy that is followed is the same as that described above for unlabeled/mislabeled specimens.
Pesticide Registration Manual: Blank Forms | US EPA Confidential Statement of Formula - EPA Form 8570-4 (pdf) Notice of Supplemental Distribution of a Registered Pesticide Product - EPA Form 8570-5 (pdf) Application for an Experimental Use Permit - EPA Form 8570-17 (pdf) Application for/Notification of State Registration of a Pesticide To Meet a Special Local Need - EPA Form 8570-25 (pdf)
Sputum Sample Collection - OSCE Guide | Geeky Medics Ensure the patient is comfortable after the procedure and check their vital signs are stable. 6. Label the specimen container at the patient's bedside with the relevant details. 7. Complete the microbiology request form, including the relevant clinical details (e.g. current symptoms, recent investigation results and past medical history). 8.
Using Our Laboratory Services - UW Laboratory Test Guide Requisition Forms. Submission of samples for laboratory testing should be accompanied by the appropriate requisition form. Laboratory procedures are performed ONLY upon a physician's written request. All verbal requests for additional testing on specimens already submitted must be followed by written confirmation within 30 days.
eOrdering with Quest Diagnostics - Elation Health To print from the Reports tab, select the desired lab order, then click "Actions" to print the requisition form or the specimen labels. The specimen labels will appear at the bottom of the requisition form and can be used with label printer paper. Printing labels follow Quest's recommendations for sizing and are 1.75" length by 0.75" height.
Clinical Laboratory Improvement Amendments (CLIA) | FDA It also helps assess patient health to make clinical decisions for patient care. The Clinical Laboratory Improvement Amendments (CLIA) regulate laboratory testing and require clinical laboratories ...
Test Request Forms for Laboratory Testing - CEDD The followings are the test request forms for Laboratory testing. Download Document(s) Form No. Item---Public Works Laboratories Test Directory (463KB)---Cement Circular ... Guidance Notes on the Use of Security Labels (1.22MB) 2322: Testing Request for Cement (297KB) 2323: Testing Request for (Clay/Concrete/Nature rock/artificial granite ...
N.C. DPH: State Laboratory of Public Health Interim Guidance Advisory: Pursuant to Session Law 2020-4 Section 4.10.(a), the Department of Health and Human Services will be requiring each person in charge of a laboratory providing diagnostic service in this State and any other health care provider licensed in this State that provides diagnostic service to report the results of all COVID-19 testing to the Department of Health and Human ...
Syphilis Treatment Follow-Up (RPR with Titer) Diagnostic Laboratories (UIDL) Test Directory 319-384-7212 (local) 1-866-844-2522 (toll free) ... Epic code: LAB8648: Order form: Laboratory Requisition: Specimen(s): Serum. Collection Medium: BD Gold SST 5 mL Vacutainer: Minimum: 5 mL serum from a BD Gold SST tube. Rejection Criteria: ... Place completed requisition into outside pocket of bag ...
Procedures for Requisition Entry and Purchase Orders Purchase Orders. Procurement receives an approved requisition and creates a purchase order based on the information entered on the requisition. The requester receives an email with a copy of the purchase order. Procurement issues the purchase order to the vendor to place the order.











Post a Comment for "40 lab requisition form with labels"