39 fda structured product labels
FDA Label Search IMPORTANT DISCLAIMER. Please be aware of the following when using information from this Web site: The drug labels and other drug-specific information on this Web site represent the most recent drug listing information companies have submitted to the Food and Drug Administration (FDA). UMLS Metathesaurus - MTHSPL (FDA Structured Product Labels) - Synopsis SPL is a document markup standard approved by Health Level Seven (HL7) and adopted by the FDA as a mechanism for exchanging medication information. Metathesaurus Scope. MTHSPL includes drug product and active substance terminology used in Structured Product Labels. MTHSPL contains approximately 158,821 drug products and 21,070 substances.
What is Structured Product Labeling? | SPL Simplified - Glemser Structured Product Labeling (SPL) is the FDA's adopted standard for communicating product and quality information. SPL includes very important information like controlled compliant components, ingredients, ingredient strengths, dosage forms, routes of administration and much more.

Fda structured product labels
Structured Product Labeling Validation Rules - Food and Drug Administration Guidance for Industry - Indexing Structured Product Labeling (Final) Guidance for Industry: Self-Identification of Generic Drug Facilities, Sites, and Organizations ... 4 Drug Labeling, Listing ... Assessing the Impact of HL7/FDA Structured Product Label (SPL) Content ... To understand the impact of SPL labels for current drug knowledge management and CPOE system implementation, this paper investigates (1) if SPL labels are sufficient as an exclusive source for drug information for e-prescribing systems today; (2) if SPL labels can be used directly in conjunction with other knowledge sources. MTHSPL (FDA Structured Product Labeling) Source Information Authority The U.S. National Library of Medicine (NLM) produces the Metathesaurus FDA Structured Product Labels (MTHSPL), which is based on the Food and Drug Administration (FDA) Structured Product Labeling (SPL). Information for this source is extracted from the NLM DailyMed Web site. Purpose
Fda structured product labels. Structure Product Labeling / Monograph (SPL-SPM) Submissions The FDA uses SPL documents to exchange information elaborating upon various product-related topics. Structured Product Monograph. In 2016, Health Canada announced that the SPL will be considered a Structured Product Monograph (SPM), and it should be submitted in an electronic format. Ensuring the same, the Agency has released guidelines and ... Indexing Structured Product Labeling | FDA Center for Drug Evaluation and Research Center for Biologics Evaluation and Research This guidance explains that FDA's Center for Drug Evaluation and Research (CDER) and Center for Biologics... What is Structured Product Labeling (SPL)?, HL7, FDA, Regulatory ... Structured Product Labeling (SPL) is a standard document approved and issued by Health Level Seven (HL7) to exchange information related to product and facility. It is used as a base for Regulatory guidance document in exchange for product labeling content. SPL ensures control over critical product information that has led to a standard for product labeling. It is adopted by the Food and Drug ... MTHSPL (FDA Structured Product Labels) - Statistics FDA Structured Product Label imprint attribute for shape text: 18077: BLA: Therapeutic Biologic Applications number for the MTHSPL drug: 15324: NDA: New Drug Application number for MTHSPL drug: 11751: DCSA: Controlled Substance Act designation code (e.g. 0,2,3n) 7193: MARKETING_EFFECTIVE_TIME_HIGH:
Structured Product Labeling (SPL) Terminology Files The NCIt-SPL terminology files provided here support the cooperative efforts of the Food and Drug Administration (FDA) and the National Cancer Institute's Thesaurus (NCIt) to develop terminology that facilitates the processing and review of SPL Terminology Files data. The efforts are described more fully on the Structured Products Labeling web ... PPTX Structured Product Labeling Overview - United States National Library ... Content of Labeling Product Data Elements Product Name Dosage Form Route of Administration Ingredient (active/inactive/adjuvant) DEA Schedule Product characteristics (color, shape, size, etc…) Packaging Marketing Information (category, status, start and end dates) Representative samples of carton/container labels Structured Product Labeling | Consumer Healthcare Products ... - CHPA In June 2009, the U.S. Food and Drug Administration (FDA) issued a guidance, "Providing Regulatory Submissions in Electronic Format — Drug Establishment Registration and Drug Listing" on its expanded requirements for submissions in the Structured Product Labeling (SPL) format. DailyMed Sep 15, 2021 · The National Library of Medicine (NLM)’s DailyMed searchable database provides the most recent labeling submitted to the Food and Drug Administration (FDA) by companies and currently in use (i.e., "in use" labeling). DailyMed contains labeling for prescription and nonprescription drugs for human and animal use, and for additional products ...
DailyMed - Download All Drug Labels Full Releases. Warning: The full human prescription and OTC archive files, dm_spl_release_human_rx.zip and dm_spl_release_human_otc.zip, are no longer available due to size considerations. Structured Product Labeling Resources | FDA Jul 28, 2022 · The Structured Product Labeling (SPL) is a document markup standard approved by Health Level Seven (HL7) and adopted by FDA as a mechanism for exchanging product and facility information. NSDE | FDA - U.S. Food and Drug Administration Mar 31, 2022 · With the exception of the billing unit data in the NSDE document, this file is generated from SPL documents sent to FDA for inclusion in the FDA Online Label Repository at labels.fda.gov. Structured Product Labeling (SPL) | Data Conversion Laboratory - DCL Structured Product Labeling (SPL) is a standard used by the FDA community to facilitate the communication of drug labeling data reliability among various groups such as the FDA, hospitals, prescribing organizations, doctors, and the general public. SPL is an HL7 and ANSI approved standard. Since 2005, the FDA CDER division has provided guidance ...
Reed Tech | Best-In-Class Information-Based Solutions and ... Build and submit UDI records electronically in Structured Product Labeling (SPL) format or seek subject-matter expertise. Manage Medical Device Product Data for UDI & Syndication Serving the medical device industry in the areas of compliance, data management and UDI regulatory requirements experience.
Structured Product Labeling Improves Detection of Drug-Intolerance Issues Introduction and Objective. The HL7 Structured Product Labeling (SPL) standard 1 implemented by the FDA uses the HL7 Reference Information Model (RIM) 2 to represent the chemical and physical nature of medical products and their safe and effective use. While not all of this content is available today, we enrich the 3704 available SPLs with knowledge from the SPL terminology sources, including ...
Package Type | FDA - U.S. Food and Drug Administration In this section: Structured Product Labeling Resources Structured Product Labeling Resources Risk Evaluation and Mitigation Strategies (REMS) SPL Resources
DailyMed - FDA Resources: SPL, Other Prescription Drug Labeling ... Structured Product Labeling (SPL) is the standard format for electronic submission of the content of labeling. For SPL resources (including industry data standards for SPL), see FDA's SPL Resources page and the "Structured Product Labeling Resources" heading on FDA's Prescription Drug Labeling Resources page.
PDF Structured Product Labeling Implementation Guide for FDA Drug ... SPL Implementation Guide for FDA Drug Establishment Registration and Drug Listing v1.0 3 Terminology: None . SPL location: This information is in the beginning of the SPL file.. XML details: The instructions at the start of SPL are the same for every SPL document (the encoding set is dependent on the character encoding used in the SPL) and are in the following form:
Quantitative Labeling of Sodium, Potassium, and Phosphorus for Human ... Individuals or entities responsible for drug product labeling are encouraged to engage with FDA for advice on specific cases. Content current as of: 09/08/2022. Regulated Product(s)
FDA SPL - Structured Product & Drug Labeling Composition Process | Reed ... Structured product labeling for both prescription and over-the-counter (OTC) drugs must incorporate an overview of the scientific information needed for the correct and effective use of the drug. The labeling is broken up into sections including explanations for use (prescription drugs) or purpose (OTC drugs), adverse effects, and more.

FDA ‘Won’t Focus’ On Enforcement Of New Label Requirements For Small Companies - Nutraceuticals ...
PDF Structured Product Labeling Implementation Guide for FDA Drug ... SPL Implementation Guide for FDA Drug Establishment Registration and Drug Listing v2.0 3 Terminology: None . SPL location: This information is in the beginning of the SPL file.. XML details: The instructions at the start of SPL are the same for every SPL document (the encoding set is dependent on the character encoding used in the SPL) and are in the following form:
SPL Xforms | FDA - U.S. Food and Drug Administration To register, please submit the following information via e-mail to spl@fda.hhs.gov: Attendee's first and last name. Name of your organization. E-mail address. Session name and date of training ...
Drug Labeling Overview - Food and Drug Administration Drug manufacturers and distributors submit documentation about their products to FDA in the Structured Product Labeling (SPL) format. The openFDA drug product labeling API returns data from this...
Structured Product Labeling - Wikipedia Structured Product Labeling ( SPL) is a Health Level Seven International (HL7) standard which defines the content of human prescription drug labeling in an XML format. [1] The "drug labeling" includes all published material accompanying a drug, such as the Prescribing Information which contains a great deal of detailed information about the drug.
IIS COVID-19 Vaccine Related Code | CDC The following vaccines and associated tradenames have been approved by the FDA under BLA License. They are listed separately because while they may represent the same formulations as the EUA authorized and labeled products listed above, the NDCs listed with the new BLA licensed tradenames in the FDA BLA approval or the FDA Structured Product Labels (SPL) are not currently being produced by the ...
A dataset of 200 structured product labels annotated for adverse drug ... The Structured Product Labels (SPLs), the documents FDA uses to exchange information about drugs and other products, were manually annotated for adverse reactions at the mention level to ...
Structured Product Labeling - Food and Drug Administration Structured Product Labeling (SPL) is a document markup standard approved by Health Level Seven (HL7) and adopted by FDA as a mechanism for exchanging product and facility information. The openFDA...
MTHSPL (FDA Structured Product Labeling) Source Information Authority The U.S. National Library of Medicine (NLM) produces the Metathesaurus FDA Structured Product Labels (MTHSPL), which is based on the Food and Drug Administration (FDA) Structured Product Labeling (SPL). Information for this source is extracted from the NLM DailyMed Web site. Purpose
Assessing the Impact of HL7/FDA Structured Product Label (SPL) Content ... To understand the impact of SPL labels for current drug knowledge management and CPOE system implementation, this paper investigates (1) if SPL labels are sufficient as an exclusive source for drug information for e-prescribing systems today; (2) if SPL labels can be used directly in conjunction with other knowledge sources.
Structured Product Labeling Validation Rules - Food and Drug Administration Guidance for Industry - Indexing Structured Product Labeling (Final) Guidance for Industry: Self-Identification of Generic Drug Facilities, Sites, and Organizations ... 4 Drug Labeling, Listing ...




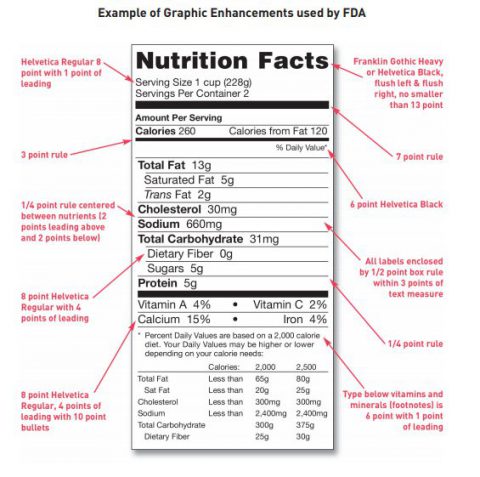





Post a Comment for "39 fda structured product labels"